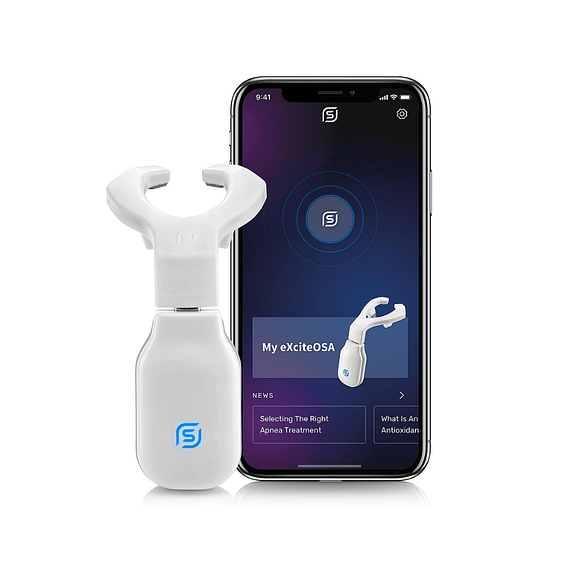
Features
- Convenient, easy to use and well tolerated daytime therapy for noticeable night-time results
- Targets a root cause of snoring and mild OSA rather than simply relieving the symptoms
- Flexible silicone mouthpiece with electrodes that positions on the tongue
- Improves muscle function of the tongue and upper airway stability through neuromuscular electrical stimulation
- Clinically proven to improve sleep quality when used for 20 minutes once daily for 6 weeks and twice weekly thereafter
This Product Includes
- Mouthpiece (flexible silicone with electrodes) with USB-C type connector*
- Control unit (With Bluetooth)
- 1 USB Cable for Control Unit Charging (15cm cable connects to UL certified USB wall charger [not provided] with 5 VDC, minimum 0.5amp output)
*Mouthpiece must be replaced approximately every 90 days requiring a separate purchase.
Please Note: eXciteOSA device is controlled by a smartphone application that can be downloaded on the Apple App Store or Google Play. See Specifications for phone requirements.
Contraindications*
Do not use eXciteOSA if you:
- Are younger than 18 years old
- Are pregnant or may be pregnant
- Have a pacemaker or implanted electrodes
- Are suffering from mouth ulcers
- Have or are suspected of having an AHI > or = to 15/hr. during a sleep study as determined by evaluation of a sleep health professional
*Safety and effectiveness in the above conditions have not been established.
Specifications
PRODUCT SPECIFICATIONS
Product Name eXciteOSA®
Manufactured By Signifier Medical Technologies
Packaging Includes Mouthpiece
1 Control Unit with Bluetooth, Includes USB-C type connector
1 USB Cable for Control Unit Charging (15cm cable connects to UL certified USB wall charger
(not provided) with 5 VDC, minimum 0.5amp output)
1 Quick Start Guide
Country of Manufacturing EU
Part Number 13010
UPC Code 060627480048
System Requirements The mobile app software can be used on iPhone 5S, iPhone 6/6 Plus, iPhone 6s/6s Plus, iPhone 7/ 7 Plus, iPhone 8/8 Plus, iPhone X, with iOS 11.0 and higher.
The mobile app software can also be used with Android devices with BLE support (Bluetooth 4.0) and Android 7.0 and above. eXciteOSA® uses Bluetooth Smart; mobile devices used must be compatible with Bluetooth Smart.
Description eXciteOSA® is the world’s first clinically proven daytime therapy for mild obstructive sleep apnea and snoring. Worn for 20 minutes, 1 time each day for 6 weeks, eXciteOSA® is focused on solving the root cause of mild OSA and snoring – the loss of muscle function of the tongue.
How eXciteOSA® Works
1. Download the eXciteOSA® app for iOS or Android directly on your phone or tablet.
2. Attach the eXciteOSA® mouthpiece to the control unit and then place into the mouth.
3. The eXciteOSA® app activates the electrodes in the mouthpiece via the control unit; tiny electrical pulses stimulate the tongue muscle and improve muscle function.
4. The eXciteOSA® app monitors patient use of the device.
Intended Use eXciteOSA® is intended for the reduction of snoring and mild obstructive sleep apnea by strengthening tongue muscles via electronical muscle stimulation.
Frequency of Use It is recommended you use eXciteOSA® once daily for a continuous 20 minutes. It can take up to 6 weeks of daily therapy before you will gain the desired improvement in your snoring and mild OSA. Once the desired results are achieved, it is recommended to have a twice-a-week regular maintenance regime. eXciteOSA® can be used long term. This is comparable to maintaining your physical fitness by undertaking regular exercise. You must replace the Mouthpiece after 90 calendar days of first use, this includes the maintenance period.
Operational Requirements Temperature +41°F to +104°F (+5°C to +40°C); Humidity 15% to 90%; Atmospheric Pressure 10.15 psi to
15.37 psi (70 kPa to 106 kPa). Operational life is 3 years.
Transport and Storage Requirements
Temperature -13°F to +158°F (-25°C to +70°C); Humidity up to 90%
Atmospheric Pressure 10.15 psi to 15.37 psi (70 kPa to 106 kPa). When the device is exposed to extreme
temperatures, please wait at least 60 minutes before attempting to use the device.
Warranty 3 months for the eXciteOSA® Mouthpiece and 1 year for the eXciteOSA® Control Unit from the date of purchase
Prescription Information Prescription required by licensed physician to purchase this item
Reimbursement Typically not reimbursable with highly variable exceptions.